38. Label-Free Detection of Virus-Membrane-Interactions
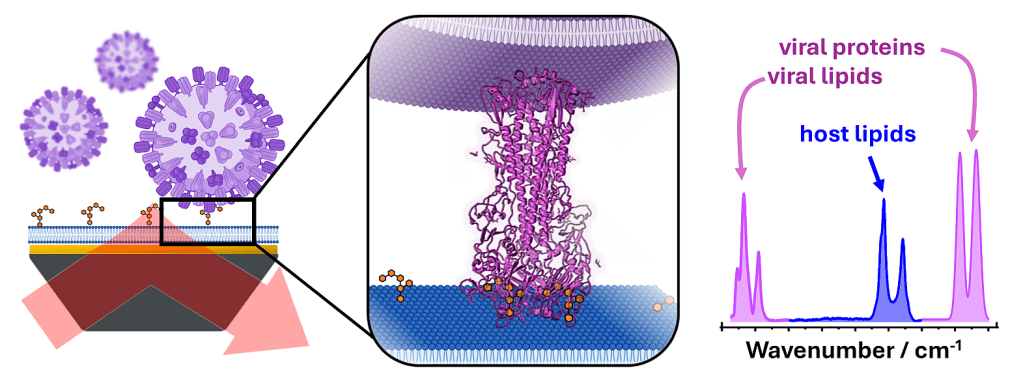
Many virus binding and fusion assays rely on the introduction of fluorescent dyes into viruses of membrane models. Combining model membranes with surface-enhanced IR absorption spectroscopy enables label-free monitoring of virus binding and fusion with molecular structure information.
Heinen et al., Angew. Chem. Int. Ed., 2025
Label-Free Detection of Virus-Membrane-Interactions Using Surface-Enhanced Infrared Absorption (SEIRA) Spectroscopy.
DOI: 10.1002/ange.202502998
33. How a potential Anticancer-Peptide interacts with Membranes

Enniatins are known as ionophores that perturb the cellular homeostasis, but have recently been re-discovered as potential anticancer drugs. Interaction with Na+ and K+ causes membrane incorporations, but Mg2+ and Ca2+ lead to abstraction as a potential mechanism of cell entry.
Gonzalez et al., ChemPlusChem, 2024
Cation Dependence of Enniatin B/Membrane-Interactions Assessed Using Surface-Enhanced Infrared Absorption (SEIRA) Spectroscopy.
DOI: 10.1002/cplu.202400159
31. Watching an Viral Proton Channel open using SEIRA spectroscopy
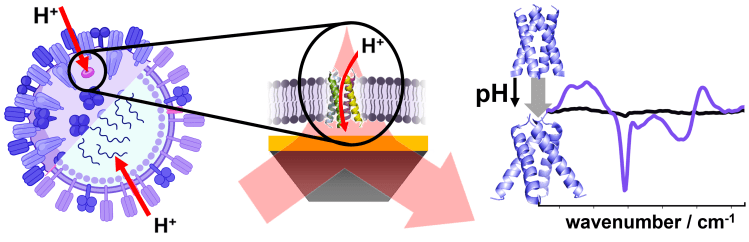
Viroporins are viral ion channels that play important role during viral infection and, thus, present important drug targets. Using SEIRA spectroscopy we monitor the opening of the Influenza A M2 proton channel in a model membrane.
Paschke et al., Angew. Chem. Int. Ed., 2023
In situ spectroscopic detection of large-scale reorientations of transmembrane helices during influenza A M2 channel opening.
DOI: 10.1002/anie.202309069
30. Review on: SEIRA spectroscopy

SEIRA spectroscopy is an important tool in the molecular analysis of surfaces. We review the basis for SEIRA, common approaches as well as important case studies in the fields of electrochemistry, trace analysis and (bio)functional investigations.
Kozuch et al., Nat. Rev. Methods Primers, 2023
Surface-enhanced infrared absorption spectroscopy.
DOI: 10.1038/s43586-023-00253-8
28. Quantifying Electrostatics at Model Membranes

We have used electrode-supported model membranes in many previous studies to control the transmembrane potential. Here we quantify the transmembrane electrostatics combining SEIRA spectroscopy and MD simulations.
Utesch et al., J. Phys. Chem. B, 2022
Potential Distribution across Model Membranes.
DOI: 10.1021/acs.jpcb.2c05372
18. Cathelicidine LL-37 – A Peptide of our innate Antimicrobial immune system

Cathelicidins are a family of host defense antimicrobial peptides in mammalian species. Among them, LL-37 is the only peptide of this family found in humans. We combine microbiology, spectroscopy and simulations to unravel its mode of action.
de Miguel Catalina & Forbrig et al., Biochemistry, 2019
The C-Terminal VPRTES Tail of LL-37 Influences the Mode of Attachment to a Lipid Bilayer and Antimicrobial Activity.
DOI: 10.1021/acs.biochem.8b01297
14. Monitoring the Catalytic Proton Translocation Across the Membrane by Respiratory Complex I
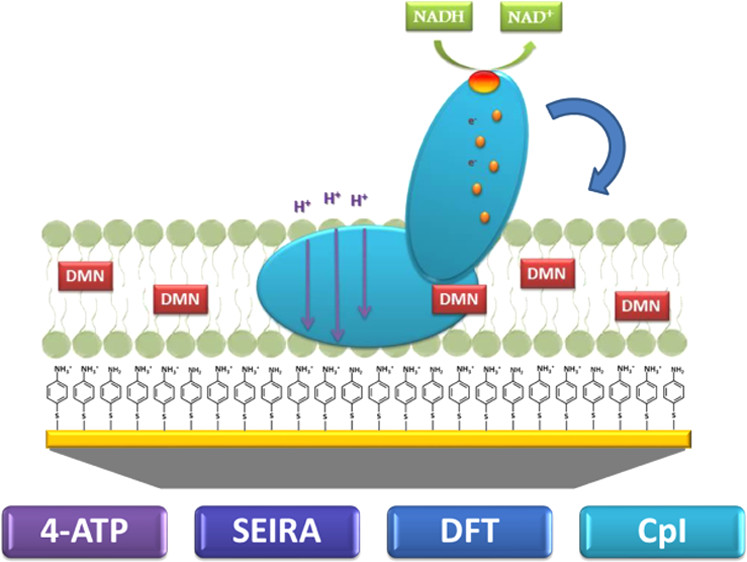
Using a pH-dependent self-assembled monolayer (SAM), based on amino thiophenol, we monitor the protein orientation-dependent directionality of the catalytic proton translocation induced by addition of NADH.
Gutierrez-Sans & Forbrig et al., Langmuir, 2018
Catalytic activity and proton translocation of reconstituted respiratory complex I monitored by surface-enhanced infrared absorption spectroscopy.
DOI: 10.1021/acs.langmuir.7b04057
12. Monitoring how Transmembrane Electrostatics reorient a-helices in Membranes
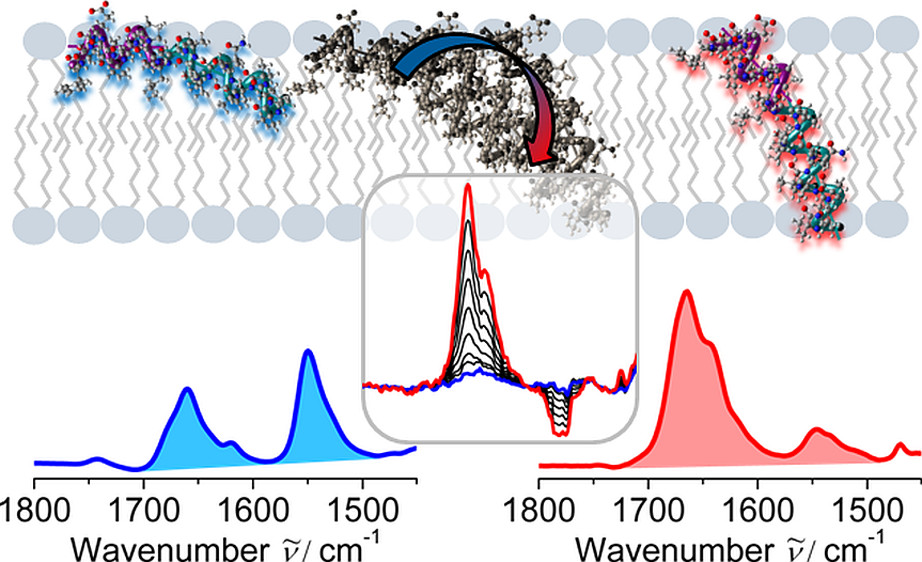
Antimicrobial peptides are the first line of defense after contact of an infectious invader, often acting via interactions with the target membrane. The transmembrane electrostatics can play a major governing role in antimicrobial action. We show, for the first time using SEIRAS, that we can control the large-scale reorientation of AMP helices to form the active ion channel form.
Forbrig et al., Langmuir, 2018
Monitoring the orientational changes of alamethicin during incorporation in bilayer lipid membranes.
DOI: 10.1021/acs.langmuir.7b04265
9. An tethered bilayer membrane system Tailored for IR Studies Membrane Proteins

Electrode-interfaced membrane systems are necessary to study and utilize membrane proteins for various biotechnological applications. However, previous IR spectro-electrochemical studies used membrane systems developed for electrochemical approaches. We construct a IR-tailored membrane system and demonstrate its applicability by monitoring the catalytic generation of the proton-motive force by cbo3 oxidase.
Wiebalck & Kozuch et al., J. Phys. Chem. B, 2016
Monitoring the transmembrane proton gradient generated by cytochrome bo3 in tethered bilayer lipid membranes using SEIRA spectroscopy.
DOI: 10.1021/acs.jpcb.6b01435
6. The mitochondrial human voltage-dependent anion channel (HVDAC) acts via deformation Its b-barrel structure

The voltage-dependent anion channel (VDAC) regulates the transfer of metabolites between the cytosol and the mitochondrium. Opening and partial closing of the channel is known to be driven by the transmembrane potentia. Our results indicate alterations of the inclination angle of the β-strands as crucial molecular events, reflecting an expansion or contraction of the β-barrel pore.
Kozuch et al., Phys. Chem. Chem. Phys., 2014
Voltage-dependent structural changes of the membrane-bound anion channel hVDAC1 probed by SEIRA and electrochemical impedance spectroscopy.
DOI: 10.1039/C4CP00167B
3. Lipid-tethered membrane systems on Plasmonic surfaces Enable IR Spectroscopy and Electrochemistry
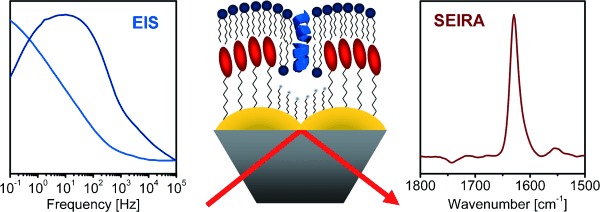
Tethered membrane systems have been routinely used in electrochemical studies. We construct for the first time a tethered membrane with insulating properties on a plasmonic, nanostructured Au electrode and demonstrate, as proof of concept, the spectroscopic and electrochemical analysis of the ion channel-forming peptide gramicidin A.
Kozuch et al., Angew. Chem. Int. Ed., 2012
Combined electrochemistry and surface-enhanced infrared absorption spectroscopy of gramicidin A incorporated into tethered bilayer lipid membranes.
DOI: 10.1002/anie.201203214